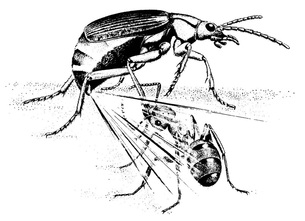
 Illustration of the Bombardier Beetle defense mechanism against predator
Illustration of the Bombardier Beetle defense mechanism against predator
The Bombardier Beetle is famous for its incredibly unique and effective defense mechanism, which they use when threatened and disturbed much like chemical warfare on an insect level. The common name Bombardier comes from the fact that the insect shoots a boiling chemical liquid from there abdomen and special glands with a 'popping. To the human ear this sounds as if there is one single 'pop' or fire of the toxin, however, after investigation it was found that the beetles actually release the toxin in pulses, almost similar to a turret gun in human terms (Dean, Aneshansley, Edgerton, & Eisner, 1990).
What is it:
The beetle's toxic liquid that it ejects is a mixture of hydrogen peroxide and hydroquinones, which are stored in separate reservoirs before being secreted by special purpose cells. When the beetle is under attack by a potential predator or feels threatened the two liquids are combined when fired through the tip of the abdomen, which can rotate 270 degrees to actually aim at their enemy. The ejection of the liquid appears as a fine mist and steam, as can be seen by the video link below (Dean, Aneshansley, Edgerton, & Eisner, 1990).
What is it:
The beetle's toxic liquid that it ejects is a mixture of hydrogen peroxide and hydroquinones, which are stored in separate reservoirs before being secreted by special purpose cells. When the beetle is under attack by a potential predator or feels threatened the two liquids are combined when fired through the tip of the abdomen, which can rotate 270 degrees to actually aim at their enemy. The ejection of the liquid appears as a fine mist and steam, as can be seen by the video link below (Dean, Aneshansley, Edgerton, & Eisner, 1990).
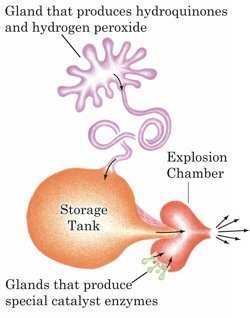
What is the chemical:
The chemical liquid released from the abdomen of the beetle has to undergo a reaction in order for the liquid to be boiling and dangerous to predators. Despite common ideas, a reaction does not occur between hydrogen peroxide and hydroquinones alone, yet a catalyst must be added to induce the heat and toxicity of the liquid. This catalyst produces the active component, p-benzoquinones, a quinone that is boiling and heated. This compound is produced by the mixture of both hydrogen peroxide and hydroquinones with an oxidation enzyme (catalyst) when ejected from the beetle. This catalyst actually starts the reaction between the two chemicals (Dean, Aneshansley, Edgerton, & Eisner, 1990).
How is it stored and fired from the beetle:
Quinones, the dangerous compounds of the liquid, are not stored in the beetle but produced rapidly from the mixture of the stored chemicals and catalyst. These two chemicals are stored in large inner chambers in different glands, with the oxidase catalyst stored in a smaller outer chamber of each gland. The larger chambers are referred to as reservoirs and the smaller outer chambers is referred to as the reaction chamber as this is where the reaction producing the boiling toxin rapidly takes place. The reservoirs have a thin cell wall surrounded by muscle tissue that is compressible while the reaction chamber is rigid with thick cell walls. A tight valve that separates the reservoir and reaction chambers in each gland are temporarily opened by muscle contractions in the reservoir releasing the liquid into the reaction chamber where the reaction and formation of the quinones are completed. This reacted liquid combination is then rapidly released in pulses with the liquid at a temperature at 100 degrees Celsius. This is why early naturalists made the observation that large beetles were hot to touch by hand. The rapid pulsing firing of the liquid by these beetles is unique to this insect in all of nature as it is something only seen in technology not animals (Dean, Aneshansley, Edgerton, & Eisner, 1990). Although it is not confirmed, research suggests that the rapid machine gun like pulses (Amato, 1990) are individual reactions and 'explosions' from the reaction of the two chemicals and the catalyst from continuous muscle contractions from the reservoirs (Dean, Aneshansley, Edgerton, & Eisner, 1990). This firing method is extremely effective in deterring predators, with the force, temperature, toxicity and noise, the Bombardier Beetle is not an insect to be messed with (Isaak, 1997).
The chemical liquid released from the abdomen of the beetle has to undergo a reaction in order for the liquid to be boiling and dangerous to predators. Despite common ideas, a reaction does not occur between hydrogen peroxide and hydroquinones alone, yet a catalyst must be added to induce the heat and toxicity of the liquid. This catalyst produces the active component, p-benzoquinones, a quinone that is boiling and heated. This compound is produced by the mixture of both hydrogen peroxide and hydroquinones with an oxidation enzyme (catalyst) when ejected from the beetle. This catalyst actually starts the reaction between the two chemicals (Dean, Aneshansley, Edgerton, & Eisner, 1990).
How is it stored and fired from the beetle:
Quinones, the dangerous compounds of the liquid, are not stored in the beetle but produced rapidly from the mixture of the stored chemicals and catalyst. These two chemicals are stored in large inner chambers in different glands, with the oxidase catalyst stored in a smaller outer chamber of each gland. The larger chambers are referred to as reservoirs and the smaller outer chambers is referred to as the reaction chamber as this is where the reaction producing the boiling toxin rapidly takes place. The reservoirs have a thin cell wall surrounded by muscle tissue that is compressible while the reaction chamber is rigid with thick cell walls. A tight valve that separates the reservoir and reaction chambers in each gland are temporarily opened by muscle contractions in the reservoir releasing the liquid into the reaction chamber where the reaction and formation of the quinones are completed. This reacted liquid combination is then rapidly released in pulses with the liquid at a temperature at 100 degrees Celsius. This is why early naturalists made the observation that large beetles were hot to touch by hand. The rapid pulsing firing of the liquid by these beetles is unique to this insect in all of nature as it is something only seen in technology not animals (Dean, Aneshansley, Edgerton, & Eisner, 1990). Although it is not confirmed, research suggests that the rapid machine gun like pulses (Amato, 1990) are individual reactions and 'explosions' from the reaction of the two chemicals and the catalyst from continuous muscle contractions from the reservoirs (Dean, Aneshansley, Edgerton, & Eisner, 1990). This firing method is extremely effective in deterring predators, with the force, temperature, toxicity and noise, the Bombardier Beetle is not an insect to be messed with (Isaak, 1997).
Image and Video Credits:
- Bombardier Beetle defense video: https://www.youtube.com/watch?v=Pib9qT-pccI
- Diagram of Bombardier Beetle glands: https://www.apologeticspress.org/apcontent.aspx?category=9&article=1113
- Illustration of Bombardier Beetle defense: http://bioinspired.sinet.ca/content/evolutionary-basis-chemical-defense-bombardier-beetles-kathryn-nagel-marc-weissburg
Alexander Kloppers
The University of Queensland, BIOL2205
The University of Queensland, BIOL2205
